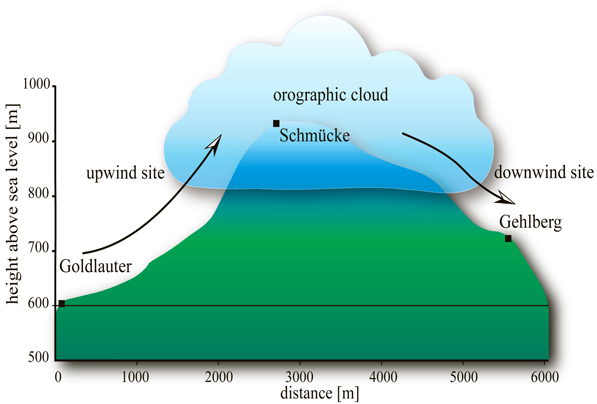
Scheme of the Schmücke campaign area with upwind site Goldlauter, in-cloud site Schmücke, and downwind site Gehlberg.

Clouds strongly impact phyical and chemical processes in the atmosphere. Studying cloud processes in a natural environment is challenging due to the altitude as well as the spatial and temporal variability of clouds. Ground-based hill-cap cloud experiments with associated upwind and downwind valley stations offer the possibility to physically and chemically characterise gas phase, aerosol particles, and cloud droplets before, during, and after the passage of an air mass through a cloud. This can help to better understand many different cloud processes. TROPOS is thus organising and leading such complex field experiments on aerosol-cloud interactions on a regular basis with international collaborations. These campaigns take place at Mount Schmücke in the Thuringian forest area, where a 20 m high measurement tower has been built for this purpose, which hosts many of the typically applied sampling techniques (Fig. 1). A multitude of measurement techniques is used to study gas phase, aerosol particles, and cloud water at the three campaign sites (Fig. 2). Interesting impacts of a cloud on physical and chemical properties of aerosol particles have been observed in these experiments:

Scheme of the Schmücke campaign area with upwind site Goldlauter, in-cloud site Schmücke, and downwind site Gehlberg.